Director of Quality Assurance
Skills
About This Role
Overview
Akura Medical, a Shifamed portfolio company, is advancing a differentiated platform for the treatment of venous thromboembolism (VTE), which affects up to 900,000 Americans each year.
The company recently received US IDE approval for its QUADRA-PE study evaluating the Katana™ Thrombectomy System in acute pulmonary embolism patients.
In parallel, Akura Medical is developing NavIQ™, a software platform that transforms CT angiograms into 3D pulmonary vascular models to enhance visualization, procedural planning, and clot assessment towards better safety, efficacy, and ease of use.
Akura has recently completed a $53 million first close of its Series C financing, led by the Qatar Investment Authority, supporting the launch of a joint venture and a new R&D and manufacturing center in Doha, Qatar.
About Shifamed
Founded in 2009 by serial entrepreneur Amr Salahieh, Shifamed LLC is a privately held medical device innovation hub focused on the development of novel medical products to address clinical needs in the rapidly evolving fields of cardiology and ophthalmology.
Description
This position is responsible for the development, implementation, and management of quality systems and practices.
This position will oversee and ensure that the development, manufacture, testing and release of products are performed according to the applicable federal and state regulations, international standards, and the Company’s Quality Policy.
Responsibilities, Skills & Hands-On Experience
- Ensure compliance with applicable Quality System Regulations and standards, including FDA QSR, ISO 13485, MDR, GMP, and Design Controls.
- Partner with R&D and Operations to establish and maintain quality requirements throughout product development, transfer, manufacturing, and post-market activities.
- Lead and maintain Design History Files (DHF), Risk Management Files, and associated quality documentation for products and processes.
- Develop and implement quality engineering activities including design verification/validation, process validation, equipment qualification, test method validation, and first article inspections.
- Support manufacturing quality activities including inspection processes, nonconformance investigations, CAPA, root cause analysis, and continuous improvement initiatives.
- Develop, maintain, and oversee manufacturing and quality documentation including procedures, inspection instructions, Lot History Records, and Bills of Materials.
- Manage supplier quality activities including supplier qualification, audits, source inspections, supplier controls, and ongoing quality performance monitoring.
- Prepare for, host, and support internal, customer, supplier, and regulatory audits and inspections.
- Coordinate quality system training and promote company-wide quality system awareness and compliance.
- Support complaint handling, MDR/vigilance reporting, field actions, and investigation of product or process failures.
- Serve as Quality representative on cross-functional project teams and act as site Management Representative and/or Quality signature authority as required.
- Lead and manage Quality Assurance resources to ensure effective execution of quality system and regulatory compliance activities.
- Other duties as assigned.
Education & Work Experience
- Bachelors or Masters in Engineering or scientific discipline, or equivalent relevant work experience
- 15+ years of Quality or Technical Management experience in the medical device industry or a related field.
- Previous direct people management experience.
- Previous experience bringing a company into regulatory compliance and approval, preferred.
- Solid knowledge of FDA regulations and interpretation of FDA regulations, guidelines, and policy statements.
- Solid knowledge of international regulations, standards, and guidelines (i.e. MDR, ISO 13485, ISO 14971, IEC 60601 and collateral standards, IEC 62304, IEC 62366, FDA guidance documents for cybersecurity).
- Ability to train others in processes and procedures.
- Excellent communications skills (both written and verbal) required.
- Ability to work effectively across functional lines to reach effective outcomes for the business.
- Ability to travel as required.
- Our salary ranges are calculated by role, level, and location.
- Please note that your position within that range will be determined by your job-related knowledge, location, skills, experience, relevant education, and training/certifications.
- NOTICE TO CANDIDATES: Please be aware that Shifamed and its portfolio companies do not conduct interviews or extend offers through mobile web chat applications.
- Please report any such occurrences to hr@shifamed.com.
Your resume, rewritten
for this exact role.
Sign up free — Base Career tailors your CV to this job description in 60 seconds.
01 / 05
Resume Tailored to This Job
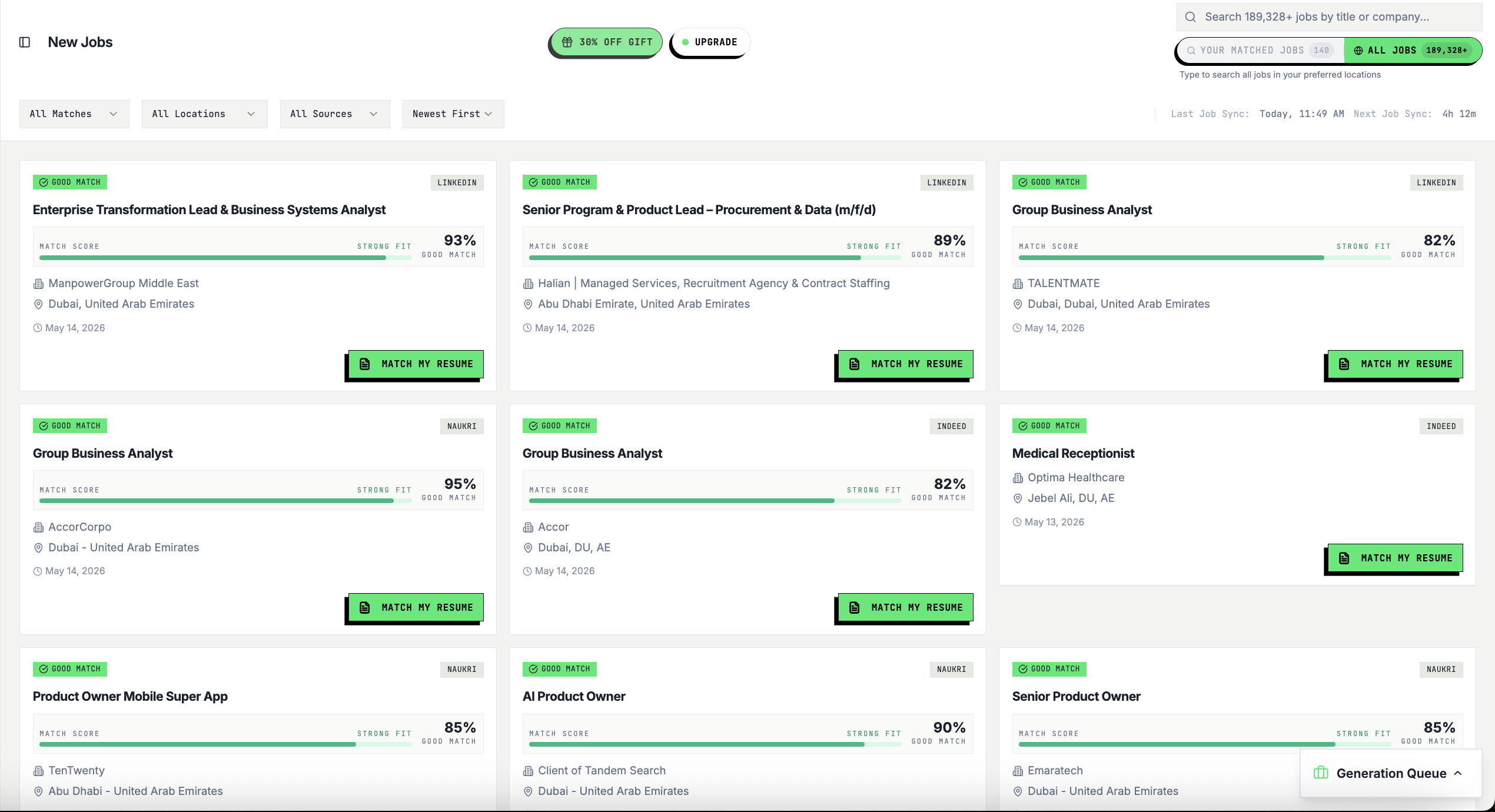
Your keywords, structure, and story — rewritten to match this exact role and pass ATS filters.
Free · No card · 60 seconds
02 / 05
Cover Letter for This Role, Done

Job-specific cover letters written in Gulf professional tone — ready in seconds, not hours.
Free · No card · 60 seconds
03 / 05
See How Well You Fit This Role
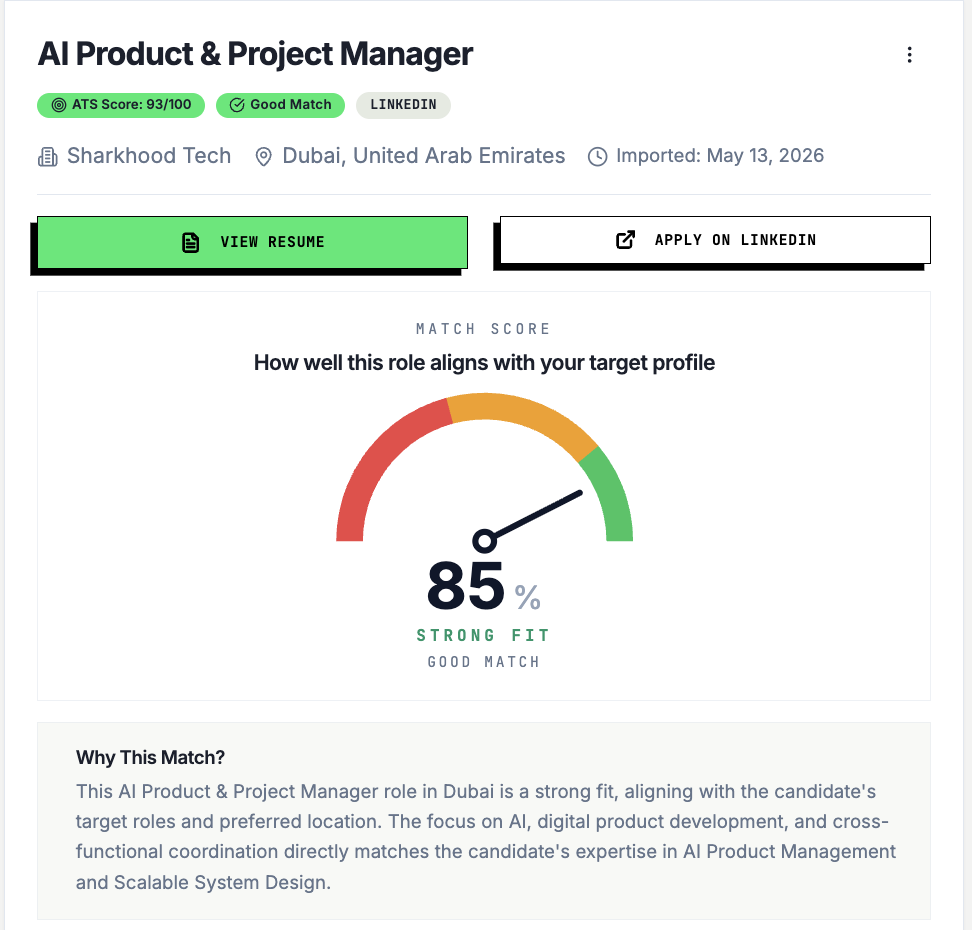
AI match score with clear reasons — know your fit before investing time in the application.
Free · No card · 60 seconds
04 / 05
Apply in One Click
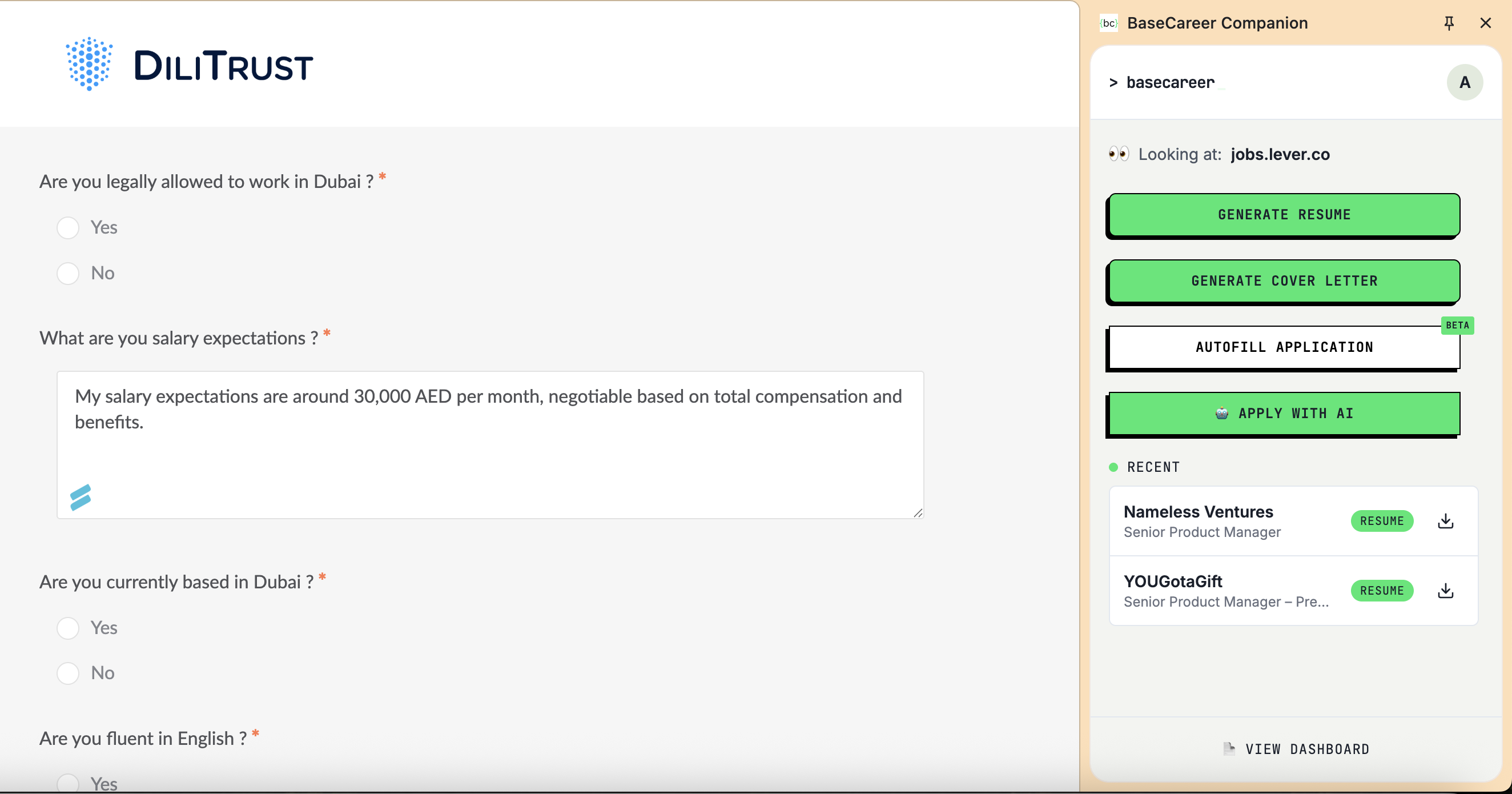
Autofill any application form on Workday, LinkedIn, Bayt, Greenhouse — with your tailored content.
Free · No card · 60 seconds
05 / 05
Track It. Follow Up at the Right Time.
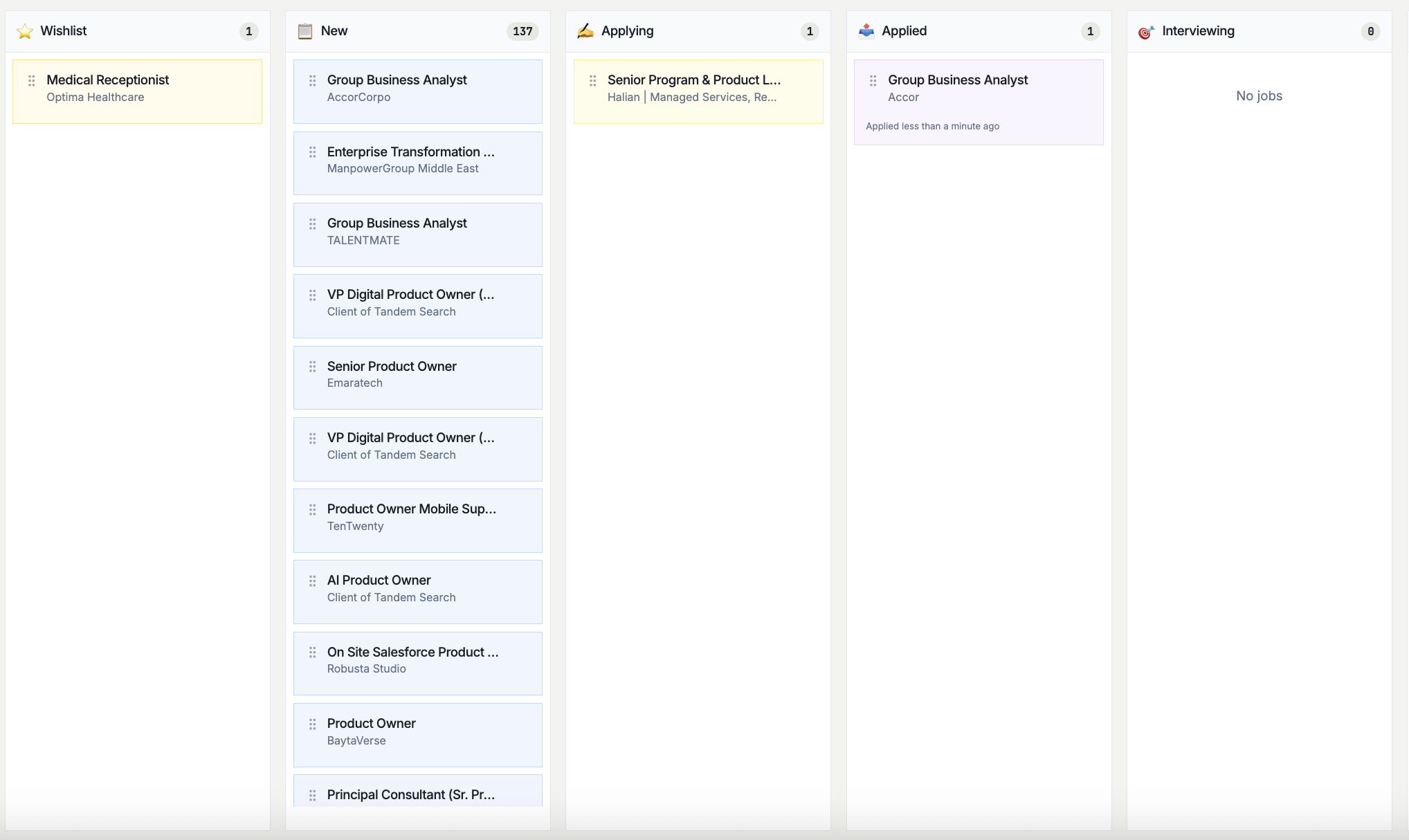
Visual pipeline for every application with AI-timed follow-up reminders so nothing slips.
Free · No card · 60 seconds
Similar Jobs
Cluster Director of Sales
Rixos · Doha
Company Description Rixos Premium Qetaifan Island North Rixos Premium Qetaifan Island North offers guests a luxurious breakaway to an island packed with entertainment and adventure. With five acres of fun-filled attracti
Skills
Director of Legal Affairs
RP International · Doha
We are looking for an experienced legal professional to join a leading multinational telecommunications company in Qatar as Director of Legal Affairs. This role will focus on providing legal and regulatory support across
Skills
Director Of Food & Beverage
The Ned Hotel · Doha
DOHA Director Of Food & BeverageFull Time SALARY RANGE CompetitiveABOUT THE NED DOHA: Housed within Doha’s iconic former Ministry of Interior building, The Ned Doha is not just a hotel — it is a refined luxury destinatio
Skills
Director of Food & Beverage
The Ned & Ned's Club · Doha
About The Ned Doha: Housed within Doha’s iconic former Ministry of Interior building, The Ned Doha is not just a hotel — it is a refined luxury destination where heritage, elegance, and contemporary sophistication come t
Skills
Director of Commercial Strategy
Mandarin Oriental Hotel Group · Doha
Mandarin Oriental, Doha is looking for a Director of Commercial Strategy to join our team. Are you a master of craft? Do you thrive in a team that succeeds together, demonstrating integrity and respect while acting respo
Skills
Director of Commercial Strategy
Mandarin Oriental · Doha
Mandarin Oriental, Doha is looking for a Director of Commercial Strategy to join our team. Are you a master of craft? Do you thrive in a team that succeeds together, demonstrating integrity and respect while acting respo
Skills
Assistant Director of Sales
ACCOR · Doha
Assist in sales activities, lead the sales team, develop business relationships, and ensure effective communication while achieving sales targets.
Skills
Assistant Director of Sales
AccorHotel · Doha
Assist in sales activities, lead the sales team, develop customer relationships, analyze market trends, and demonstrate strong leadership and communication skills.
Skills
Assistant Director of Sales
Accor · Doha
Company Description La Cigale Hotel Managed by Accor is a prestigious five-star luxury hotel situated in the dynamic center of Doha, Qatar. Recognized as a prominent landmark for both business and leisure travelers, the
Skills
Professionals hired via Base Career
“I kept getting rejections from London. Base Career rewrote my CV for Dubai, and I landed Emirates in 3 weeks.”
Sarah M. · Marketing Manager
🇬🇧 UK → 🇦🇪 Dubai
“50 applications in Canada, zero replies. Base Career tailored my resume for Riyadh and I got 4 interviews within a month.”
James T. · Software Engineer
🇨🇦 Canada → 🇸🇦 Riyadh
“The cover letters matched Gulf tone immediately. I got hired by a semi-government team in Doha on my first round.”
Maya R. · Product Manager
🇺🇸 USA → 🇶🇦 Doha
“As an expat I had no idea how Gulf CVs work. Base Career nailed it. Offer from a Big 4 in Abu Dhabi in 6 weeks.”
Priya K. · Finance Analyst
🇮🇳 India → 🇦🇪 Abu Dhabi
2.2K+
Cover Letters & Follow-ups
1.8K+
Resumes Tailored
190.5K+
Jobs Tracked
Trusted by professionals at
Stop applying blindly.
Start getting hired.
Base Career automates the hardest parts of job searching — apply smarter, not harder.
AI Resume in 60s
Your resume rewritten for this exact role using the job description as the brief.
ATS-Optimized
Get past automated screening filters with the right keywords matched to each job.
Application Tracker
Track every job, follow-up, and interview in one visual kanban board.
Free plan · No credit card required